Vyvanse vs Adderall is one of the most searched medication comparisons among patients, caregivers, and clinicians evaluating ADHD treatment. Both are Schedule II central nervous system stimulants that increase dopamine and norepinephrine, but they differ meaningfully in chemistry, duration, and misuse potential.
Vyvanse (lisdexamfetamine) is a prodrug that only becomes active after metabolic conversion in the body. Adderall contains mixed amphetamine salts available in immediate-release (IR) or extended-release (XR) form.
Vyvanse lasts up to 13 to 14 hours per dose. Adderall IR lasts 4 to 6 hours; Adderall XR extends coverage to approximately 12 hours.
Choosing between them depends on a patient’s symptom pattern, medical history, and risk factors. Only a licensed prescriber can determine which medication is clinically appropriate.
Key Highlights
- Vyvanse and Adderall are both Schedule II CNS stimulants FDA-approved to treat ADHD in adults and children aged 6 and older.
- According to SAMHSA’s 2021 National Survey on Drug Use and Health, approximately 5.1 million Americans aged 12 or older misused prescription stimulants in the past year.
- Vyvanse is a prodrug, meaning its misuse potential is structurally lower because it must undergo enzymatic conversion before becoming pharmacologically active.
- Adderall holds FDA approval for narcolepsy; Vyvanse holds an additional FDA approval for moderate-to-severe binge eating disorder (BED) in adults.
- Both medications carry a black box warning for cardiovascular risk and dependence potential and should never be used without medical supervision.
What Are Vyvanse and Adderall?
Vyvanse and Adderall are prescription amphetamine-class medications used primarily to treat attention-deficit hyperactivity disorder. Both are classified as Schedule II controlled substances by the U.S. Drug Enforcement Administration, reflecting their recognized potential for dependence. They are among the most commonly prescribed ADHD drugs in the United States for both pediatric and adult patients.
What Is Vyvanse?
Vyvanse is the brand name for lisdexamfetamine dimesylate, a prodrug that is pharmacologically inactive until it is metabolized by red blood cell enzymes into the active d-amphetamine. The FDA approved Vyvanse in 2007 for ADHD and later expanded its approval to include moderate-to-severe binge eating disorder in adults. Its prodrug design produces a gradual onset and a long, smooth duration of effect that distinguishes it from other amphetamines.
What Is Adderall?
Adderall is the brand name for a combination of amphetamine and dextroamphetamine, commonly called mixed amphetamine salts. It was FDA-approved in 1996 for ADHD treatment and also carries approval for narcolepsy management. Adderall IR is immediately bioavailable after absorption and begins working within 30 to 60 minutes. Adderall XR uses a dual-bead system to release half the dose immediately and the other half over several hours.
How Vyvanse and Adderall Work
Both medications increase the synaptic availability of dopamine and norepinephrine in the central nervous system by blocking reuptake and triggering release from presynaptic storage sites. For individuals with ADHD, this neurochemical effect improves attention, impulse control, and executive function. Understanding why stimulants calm people with ADHD, rather than overstimulating them, provides important clinical context when comparing both drugs at therapeutic doses.
Adderall enters the bloodstream in its active form immediately after absorption, producing a faster and more pronounced onset. Vyvanse must first be converted by enzymes in red blood cells, which delays activation by approximately 1 to 2 hours. Once converted, lisdexamfetamine releases d-amphetamine gradually, reducing sharp plasma peaks and steep drops. The underlying neurochemical mechanism is similar in both drugs after conversion, but the delivery timing differs considerably.
FDA-Approved Uses: Vyvanse vs Adderall
The FDA-approved indications for these two medications differ in ways that influence which drug a physician is most likely to prescribe for a given patient. Adderall IR is approved for ADHD in adults and in children as young as age 3. Adderall XR is approved for ADHD in adults and children aged 6 and older. Both IR and XR formulations are also approved to manage narcolepsy in patients aged 6 and older.
Vyvanse is approved for ADHD in adults and children aged 6 and older. It is also the only stimulant FDA-approved to treat moderate-to-severe binge eating disorder in adults aged 18 and older. Physicians treating patients with comorbid ADHD and BED often have a clear clinical reason to prefer Vyvanse over other stimulants. Neither medication is approved for anxiety, depression, or weight loss, though off-label use occurs outside evidence-based prescribing standards.
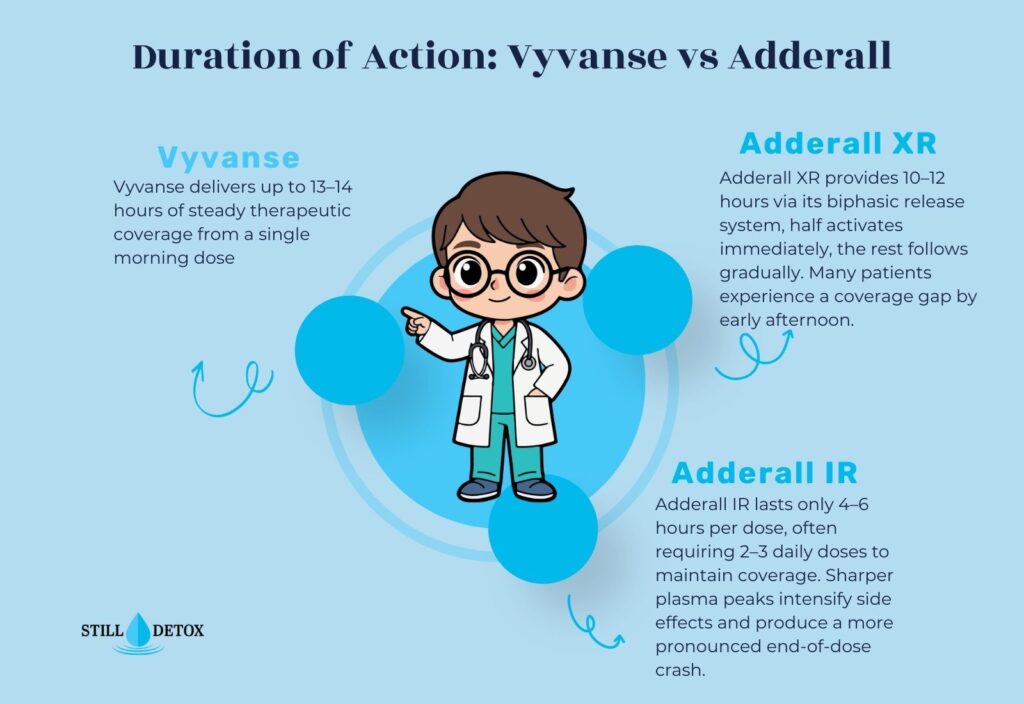
Duration of Action: How Long Do They Last?
Duration is one of the most clinically significant differences when comparing Vyvanse vs Adderall. Vyvanse provides approximately 13 to 14 hours of therapeutic coverage with a single morning dose, making it one of the longest-acting stimulants currently available. This extended, steady effect minimizes the symptom rebound many patients experience with shorter-duration medications.
Knowing how long Vyvanse stays in your system matters for understanding drug clearance, timing of side effects like insomnia, and drug testing windows. Adderall IR lasts only 4 to 6 hours, often requiring two or three doses throughout the day to maintain adequate coverage. Adderall XR extends coverage to roughly 10 to 12 hours via its biphasic release mechanism. Understanding how long Adderall stays in your system is equally important for dosing scheduling, sleep quality, and managing pharmacokinetic interactions with other substances.
Vyvanse vs Adderall Dosage Comparison
Doses are not interchangeable milligram-for-milligram between these two medications due to differences in bioavailability and conversion efficiency. A rough clinical equivalence sometimes cited is that Vyvanse 30 mg corresponds approximately to Adderall XR 10 mg, though individual patient responses vary and physician titration is always required.
| Vyvanse | Adderall IR | Adderall XR | |
|---|---|---|---|
| ADHD starting dose (children 6-17) | 30 mg once daily | 5-10 mg once daily | 5-10 mg once daily |
| ADHD max dose (children) | 70 mg/day | 40 mg/day | 30 mg/day |
| ADHD starting dose (adults) | 30 mg once daily | 5 mg once or twice daily | 20 mg once daily |
| ADHD max dose (adults) | 70 mg/day | 40 mg/day | 40 mg/day |
| BED dose (adults) | 50-70 mg once daily | Not approved | Not approved |
| Titration intervals | Weekly (10-20 mg steps) | Weekly (5 mg steps) | Weekly (5-10 mg steps) |
| Minimum approved age | 6 years | 3 years | 6 years |
Side Effects: Vyvanse vs Adderall
Vyvanse and Adderall share a largely overlapping side effect profile because both ultimately deliver d-amphetamine to the central nervous system. Adderall IR produces sharper peaks in plasma amphetamine concentration, which can intensify side effects during peak activity and cause a more pronounced crash as the dose wears off. Vyvanse’s gradual activation typically results in a smoother onset and offset with less end-of-dose rebound for most patients.
Both medications carry FDA black box warnings for cardiovascular risk and high dependence potential. Patients with structural heart abnormalities, uncontrolled hypertension, or a history of psychosis require thorough physician evaluation before starting either drug. Research confirms that Adderall can cause psychosis in vulnerable individuals, particularly at high doses or with prolonged use, and this risk applies broadly to all amphetamine-class medications, including Vyvanse.
| Side Effect | Vyvanse | Adderall IR | Adderall XR |
| Decreased appetite | Very common | Very common | Very common |
| Insomnia | Common | Common | Common |
| Dry mouth | Common | Common | Common |
| Headache | Common | Common | Common |
| Elevated heart rate | Common | Common | Common |
| Elevated blood pressure | Common | More pronounced | Common |
| Irritability/mood changes | Less pronounced | More pronounced | Moderate |
| End-of-dose crash | Minimal | Pronounced | Moderate |
| Nausea/stomach pain | Common | Common | Common |
| Weight loss | Common | Common | Common |
Is Vyvanse Less Addictive Than Adderall?
Vyvanse carries a structurally lower abuse liability than conventional amphetamines because its prodrug design prevents activation through alternative routes of administration. Snorting or injecting lisdexamfetamine does not produce the rapid dopamine surge that drives stimulant misuse, because enzymatic metabolism is still required for activation. This pharmacological barrier was a key consideration in the FDA’s assessment of Vyvanse’s relative abuse potential during the approval process.
Both drugs remain Schedule II controlled substances, meaning both carry clinically significant dependence risk in any population. Questions about whether Adderall is addictive apply equally to Vyvanse when either medication is taken in doses higher than prescribed or used without a genuine ADHD diagnosis. Heavy amphetamine use of any kind, including prescription stimulants, can result in tolerance, physical dependence, and a withdrawal syndrome upon abrupt cessation. Anyone using either medication outside of medical supervision should seek professional evaluation immediately.
Vyvanse vs Adderall for Weight Loss
Both medications suppress appetite as a pharmacological side effect, which has generated interest in their use for weight management. Neither Vyvanse nor Adderall is FDA-approved for weight loss, and using either drug for this purpose without a clinical diagnosis constitutes misuse of a Schedule II controlled substance. Appetite suppression in both drugs is a secondary effect, not a primary therapeutic mechanism.
Vyvanse is specifically FDA-approved for moderate-to-severe binge eating disorder, where it helps reduce the frequency and severity of binge episodes per week. This approval is rooted in clinical trial data demonstrating efficacy for BED, not for general weight reduction. Physicians prescribing Vyvanse for BED do so within a formal diagnostic framework with ongoing monitoring and follow-up. Weight changes observed on both medications reflect reduced caloric intake driven by appetite suppression and should be openly discussed with a prescribing physician.
Which Is Stronger: Vyvanse or Adderall?
Comparing the strength between Vyvanse and Adderall depends on which clinical outcome is being evaluated, and there is no single universal answer. Milligram for milligram, Adderall IR produces a faster and more intense peak stimulant effect because it is immediately bioavailable after ingestion. This acute intensity is what many patients describe when they say Adderall “feels stronger” at equivalent doses.
In terms of sustained daily therapeutic effect, Vyvanse generally provides longer and more stable coverage than Adderall XR for many patients. For individuals who need consistent all-day symptom management without multiple dosing, Vyvanse can feel more effective even if its peak intensity is lower. Prescribers use clinical observation and patient-reported outcomes to guide dosage decisions rather than fixed potency conversion ratios.
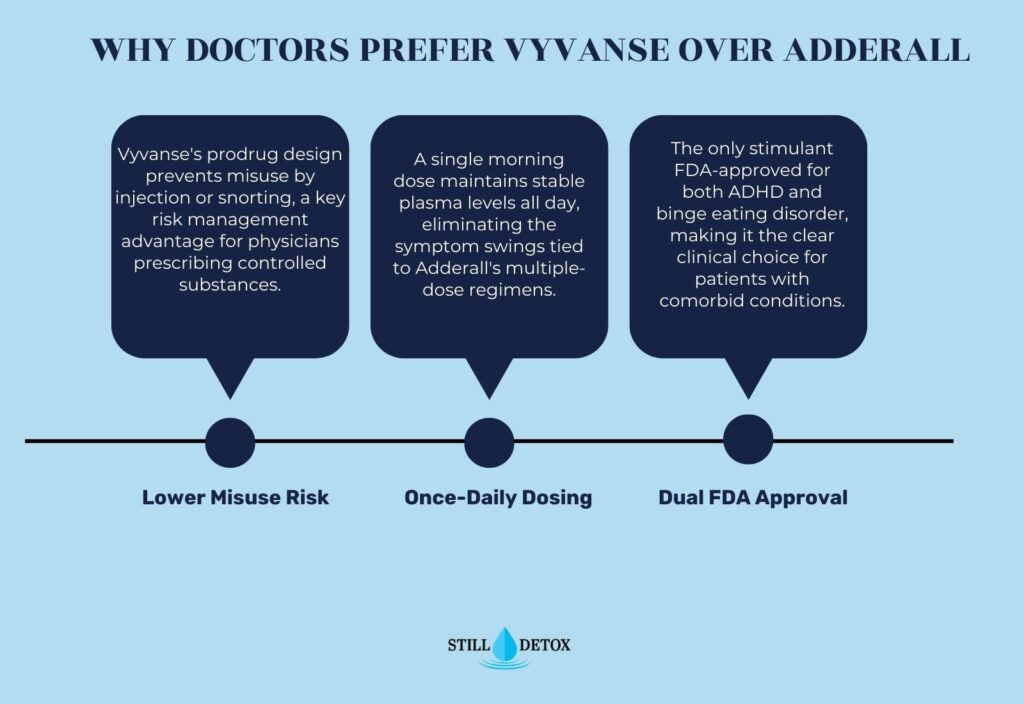
Why Do Doctors Prefer Vyvanse Over Adderall?
Multiple clinical factors drive physician preference toward Vyvanse for many ADHD patients. Its prodrug design lowers misuse liability, which is a meaningful risk management consideration for physicians prescribing controlled substances across diverse patient populations. Once-daily dosing with stable plasma levels also reduces the symptom fluctuations and behavioral variability associated with Adderall’s multiple-dose or biphasic-release regimens.
Vyvanse’s dual FDA approval for both ADHD and binge eating disorder makes it the clinically superior choice for patients presenting with comorbid conditions. The absence of a feasible alternative route for misuse simplifies risk management in patients with a history of substance use. That said, Adderall remains the preferred option when a faster onset is clinically necessary, when patients are under age 6, or when an individual has already achieved stable symptom control on existing Adderall therapy.
Why Do People Switch from Adderall to Vyvanse?
Patients commonly switch from Adderall to Vyvanse due to dissatisfaction with inconsistent daily coverage, pronounced afternoon symptom crashes, or mood instability tied to sharper pharmacokinetic peaks. Adderall XR may lose effectiveness by early afternoon in many patients, creating a coverage gap that interferes with work, school, or evening responsibilities. Vyvanse’s longer, more consistent duration directly addresses this problem for a large subset of patients.
Mood-related side effects are another significant driver of the switch. Adderall’s peaks and troughs in plasma concentration can produce emotional dysregulation, irritability, or rebound anxiety in some individuals. Vyvanse tends to produce a more stable effect throughout the day because its activation is gradual and its plasma levels remain more consistent. The widespread Adderall shortage that began in 2022 also prompted a significant number of patients to transition to Vyvanse or its now-available generic, lisdexamfetamine.
Vyvanse vs Adderall vs Ritalin
Ritalin (methylphenidate) belongs to a chemically distinct class of stimulants compared to both Vyvanse and Adderall, even though all three treat ADHD effectively in many patients. Methylphenidate primarily blocks dopamine and norepinephrine reuptake without significantly triggering release from presynaptic storage sites, the way amphetamines do. This mechanistic difference can produce different response profiles, side effect patterns, and misuse potential across patient populations.
Ritalin’s standard form is short-acting, lasting approximately 3 to 5 hours. Longer-acting methylphenidate formulations such as Concerta offer coverage more comparable to Adderall XR in terms of duration. For patients seeking once-daily dosing from the methylphenidate class, a comparison of Concerta vs Vyvanse offers useful context on how these two once-daily options differ. Physicians typically trial one stimulant class before switching to the other when initial therapy fails to achieve an adequate clinical response.
Can You Take Vyvanse and Adderall Together?
Combining Vyvanse and Adderall is not clinically recommended and should never be attempted without explicit physician direction. Because both are CNS stimulants with overlapping mechanisms, co-administration significantly elevates the risk of cardiovascular events, including dangerously elevated heart rate, high blood pressure, and arrhythmia. Excessive dopaminergic stimulation from both drugs simultaneously can also trigger anxiety, paranoia, or stimulant-induced psychosis.
No FDA-approved clinical protocol recommends prescribing both medications concurrently to the same patient. If a current stimulant is not providing sufficient symptom control, the appropriate response is a supervised dose adjustment or a formally guided medication change under physician oversight. Self-medicating with both simultaneously, regardless of intent, constitutes dangerous misuse of Schedule II controlled substances.
When Stimulant Use Becomes a Problem
Both Vyvanse and Adderall carry genuine potential for misuse, dependence, and addiction when used outside of supervised medical care. Common warning signs of stimulant misuse include taking doses higher than prescribed, using someone else’s prescription, extending waking hours artificially with stimulants, or feeling psychologically unable to function without the medication. These patterns warrant prompt professional clinical evaluation.
Amphetamine addiction treatment is evidence-based and effective for individuals who have developed dependence on prescription stimulants. Comprehensive Adderall addiction treatment typically begins with medically supervised detox followed by behavioral therapies, including cognitive behavioral therapy and motivational interviewing. Recovery from prescription stimulant use disorder is achievable with the right clinical support and a structured long-term plan.
Frequently Asked Questions
Is Vyvanse Better for You Than Adderall?
Neither medication is universally better. Vyvanse offers a smoother, longer-lasting effect with a structurally lower misuse potential due to its prodrug design. Adderall provides a faster onset and greater flexibility in dosing forms, including immediate-release options. A prescribing physician determines which is most appropriate based on an individual patient’s clinical history, age, and symptom profile.
Why Do Doctors Prefer Vyvanse Over Adderall?
Physicians often prefer Vyvanse for its lower abuse liability, consistent once-daily dosing, stable plasma levels throughout the day, and dual FDA approval for ADHD and binge eating disorder. Its prodrug structure prevents misuse through snorting or injection, which is a meaningful risk management advantage in clinical practice.
Why Do People Switch from Adderall to Vyvanse?
The most common reasons include end-of-dose crashes with Adderall, inconsistent symptom coverage throughout the day, mood instability linked to sharper plasma peaks and troughs, and the ongoing Adderall shortage that began in 2022. Many patients and physicians find Vyvanse’s once-daily, stable pharmacokinetic profile better suited to sustained daily functioning.
Why Is Vyvanse So Much Better for Some Patients?
Patients who report Vyvanse as dramatically better than Adderall typically benefit from its 13 to 14-hour coverage window and its gradual, consistent activation pattern. Fewer plasma peaks and crashes translate to more stable attention, mood, and energy across the full day rather than only during peak medication hours.
Is Vyvanse Extended Release?
Yes. Vyvanse functions as an extended-release stimulant by design, as it is a prodrug requiring enzymatic conversion before activation. This conversion process inherently creates a slow, sustained release of d-amphetamine with no immediate-release version available or possible within the same compound.
References
- Substance Abuse and Mental Health Services Administration. (2022). Key substance use and mental health indicators in the United States: Results from the 2021 National Survey on Drug Use and Health. https://www.samhsa.gov/data/sites/default/files/reports/rpt39443/2021NSDUHFFRRev010323.pdf
- U.S. Food and Drug Administration. (2023). Vyvanse (lisdexamfetamine dimesylate) prescribing information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/021977s047lbl.pdf
- U.S. Food and Drug Administration. (2013). Adderall XR (mixed amphetamine salts extended release) prescribing information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2013/021303s026lbl.pdf
- Biederman, J., Krishnan, S., Zhang, Y., McGough, J. J., & Findling, R. L. (2007). Efficacy and tolerability of lisdexamfetamine dimesylate (NRP-104) in children with attention-deficit/hyperactivity disorder. Pediatrics, 119(3), e745-e755.
- Coghill, D. R., Caballero, B., Sorooshian, S., & Civil, R. (2014). A systematic review of the safety of lisdexamfetamine dimesylate in adult patients with attention-deficit hyperactivity disorder. CNS Drugs, 28(6), 497-511.
- Martin, D., et al. (2023). Amphetamine. In StatPearls. StatPearls Publishing.