The M365 pill is a white oblong tablet containing hydrocodone bitartrate 5 mg and acetaminophen 325 mg, manufactured by Mallinckrodt Pharmaceuticals.
Federal law classifies it as a Schedule II controlled substance due to its high potential for opioid use disorder and physical dependence. Counterfeiters replicate the M365 imprint with industrial pill presses and load fake tablets with fentanyl, carfentanil, or nitazenes at doses that cause fatal respiratory depression.
Identifying the authentic pill, understanding how it works, and recognizing early signs of hydrocodone dependence can prevent a prescription from becoming a life-threatening problem.
Key Takeaways
- The M365 pill contains hydrocodone bitartrate 5 mg and acetaminophen 325 mg; it is the generic equivalent of Norco 5/325, not Vicodin, which contained a different acetaminophen dose of 300 mg per tablet.
- The DEA reclassified all hydrocodone combination products, including the M365, from Schedule III to Schedule II effective October 6, 2014, because adding acetaminophen does not reduce hydrocodone’s high abuse potential.
- According to the 2023 National Survey on Drug Use and Health (SAMHSA), 3.4 million individuals reported misusing hydrocodone products in the past year, down from a peak of 7.2 million in 2015.
- The DEA confirmed that approximately 3 in 10 counterfeit pills seized in 2025 contained a potentially lethal dose of fentanyl, meaning no unverified M365 tablet is safe to consume.
- Acetaminophen hepatotoxicity develops when total daily intake exceeds 4,000 mg; at 325 mg per tablet, exceeding 12 M365 tablets within 24 hours crosses the FDA boxed warning threshold.
What Is the M365 Pill?
The M365 pill is a Schedule II opioid analgesic manufactured by Mallinckrodt Pharmaceuticals, combining hydrocodone bitartrate 5 mg and acetaminophen 325 mg in each white oblong tablet to treat moderate to moderately severe acute pain.
Active Ingredients and Manufacturer
Hydrocodone is a semi-synthetic mu-opioid receptor agonist derived from codeine. The “M” imprint identifies Mallinckrodt Pharmaceuticals as the manufacturer; “365” specifies this exact formulation and distinguishes it from other Mallinckrodt hydrocodone products with different milligram strengths. Acetaminophen contributes analgesia through COX enzyme inhibition in the central nervous system, independent of opioid receptor binding.
Each M365 tablet contains:
- Hydrocodone bitartrate 5 mg: the opioid analgesic responsible for pain relief, euphoria, and the primary driver of addiction risk
- Acetaminophen 325 mg: a non-opioid analgesic and antipyretic that amplifies hydrocodone’s pain-relief effect through a separate, non-receptor-binding central mechanism
- Inactive ingredients: crospovidone, magnesium stearate, microcrystalline cellulose, povidone, pregelatinized starch, silicon dioxide, and stearic acid
Brand Name Equivalents and Generic Market
The M365 is the generic equivalent of Norco 5/325. It is not equivalent to Vicodin, which contained 300 mg of acetaminophen rather than 325 mg. Norco, Vicodin, and Lortab have all been discontinued as branded products; only generic formulations under different imprint codes now remain.
Current generic equivalents for hydrocodone 5 mg/acetaminophen 325 mg include:
- IP109: manufactured by Amneal Pharmaceuticals, white oblong capsule-shaped tablet
- T257: manufactured by Camber Pharmaceuticals, white oblong capsule-shaped tablet
Schedule II Classification and Black Box Warning
The DEA reclassified all hydrocodone combination products from Schedule III to Schedule II effective October 6, 2014, after determining that adding acetaminophen does not reduce hydrocodone’s high abuse potential or severe dependence risk. Schedule II status prohibits prescription refills; each dispensing event requires a new written or electronic prescription from a licensed prescriber.
The FDA applies a black box warning to the M365 pill citing these specific risks:
- Addiction, abuse, and misuse that can lead to overdose and death
- Life-threatening respiratory depression, particularly within the first 24 to 72 hours of initiation or dose increase
- Neonatal opioid withdrawal syndrome in newborns exposed to hydrocodone during pregnancy
- Fatal drug interactions with benzodiazepines, alcohol, gabapentinoids, and other CNS depressants
M365 Pill 2025 Safety Updates
Hydrocodone Bitartrate 7.5 mg / Acetaminophen 325 mg
Toxic Leukoencephalopathy Warning (July 2025)
The FDA updated opioid labels to include warnings about Toxic Leukoencephalopathy. This is a rare but severe neurological disorder that has been linked to opioid use, particularly following instances of overdose.
Gabapentinoid Interactions
Specific warnings now address the co-administration of the M365 pill with gabapentinoids (e.g., Neurontin or Lyrica). Evidence shows a significantly heightened risk of severe respiratory depression when these medications are taken together.
Anaphylaxis Evaluation (September 2025)
As of late 2025, the FDA began a specific signal evaluation for all acetaminophen-containing products. This follows new clinical reports of severe anaphylactic (allergic) reactions in patients previously thought to have no known allergies to the drug components.
Note: This information reflects 2025 regulatory shifts. Consult a healthcare provider for personalized medical advice.
How the M365 Pill Affects the Brain and Body
The M365 pill produces analgesia through two independent molecular mechanisms that generate greater pain relief than either compound achieves alone.
Mu-Opioid Receptor Agonism and Dopaminergic Reinforcement
Hydrocodone bitartrate binds mu-opioid receptors throughout the brain and spinal cord, activating Gαi/o proteins that suppress adenylyl cyclase activity and inhibit voltage-gated calcium channels. Simultaneous potassium channel activation produces membrane hyperpolarization in dorsal horn neurons. This sequence decreases substance P release from primary afferents and reduces ascending pain signal transmission to the cortex.
Mu-opioid receptor activation in the limbic system releases dopamine into the nucleus accumbens, producing euphoria alongside analgesia. Repeated activation progressively downregulates mu-opioid receptor density through internalization, requiring escalating M365 doses to achieve equivalent effects. This receptor downregulation is the core neurological basis of hydrocodone use disorder.
CYP2D6 Metabolism and Pharmacogenomic Variability
The liver converts hydrocodone to hydromorphone via CYP2D6 O-demethylation, producing an active metabolite with approximately 30 times higher mu-opioid receptor affinity than the parent compound. This metabolite is the primary source of both analgesic effect and overdose risk. A secondary CYP3A4 N-demethylation pathway produces norhydrocodone, which is pharmacologically inactive.
Individuals with CYP2D6 ultra-rapid metabolizer phenotypes generate hydromorphone faster and at higher concentrations, increasing respiratory depression risk at standard prescribed doses. CYP3A4 inhibitors including ketoconazole and grapefruit juice shunt metabolic flux toward CYP2D6, elevating hydromorphone levels unpredictably. This pharmacogenomic variability explains why identical M365 doses produce dramatically different clinical outcomes across patients.
Acetaminophen’s Complementary Analgesic Mechanism
Acetaminophen inhibits cyclooxygenase (COX) enzyme activity in the central nervous system, reducing prostaglandin E2 synthesis and producing analgesia and antipyresis through pathways that do not involve opioid receptor binding. This non-opioid mechanism targets pain pathways that hydrocodone cannot address, creating genuine analgesic synergy rather than simple dose addition.
Acetaminophen hepatotoxicity becomes clinically significant when total daily intake exceeds 4,000 mg. At 325 mg per M365 tablet, exceeding 12 tablets within 24 hours crosses the FDA boxed warning threshold. Concurrent use of acetaminophen-containing cold medicines, sleep aids, or other analgesics accumulates toward this ceiling without patients recognizing the combined exposure.
How to Identify the M365 Pill
The M365 pill has precise physical characteristics defined by FDA manufacturing specifications; any deviation from these characteristics indicates a potentially counterfeit tablet.
Physical Description and Imprint Characteristics
The authentic M365 pill is white, capsule-shaped (oblong), approximately 15 mm in length, with a scored center line dividing the tablet face into two equal halves. The “M365” imprint appears on one flat surface in uniform, clean characters with consistent depth and centered positioning. The reverse face carries only the score line. Every authentic Mallinckrodt tablet is manufactured to FDA dimensional tolerances, producing uniform weight, density, and surface hardness across all pills in a production batch.
The M365 is frequently mislabeled “oval” in online searches, including in the existing article URL. Oblong and oval are not interchangeable pill shape descriptors. An oblong tablet has an elongated rectangular profile with rounded ends, similar to a large capsule. An oval tablet is wider at the center and tapers at both ends, resembling an egg. The FDA’s official drug database classifies the M365 as “Capsule/Oblong,” not oval. Counterfeit tablets pressed from imprecise molds sometimes deviate toward an egg shape, making this dimensional distinction a visual identification signal.
M365 and Mallinckrodt Hydrocodone Variant Comparison
The M365 is one of three Mallinckrodt hydrocodone/acetaminophen tablets that differ only in hydrocodone dose and shape. The 10 mg variant is the M367 pill; the comparison table below covers all five major generic equivalents currently dispensed in US pharmacies. All five imprints are Schedule II controlled substances regardless of hydrocodone milligram strength.
| Imprint | Manufacturer | Hydrocodone | Acetaminophen | Shape | Schedule |
|---|---|---|---|---|---|
| M365 | Mallinckrodt | 5 mg | 325 mg | White oblong | II |
| M366 | Mallinckrodt | 7.5 mg | 325 mg | White oval | II |
| M367 | Mallinckrodt | 10 mg | 325 mg | White oblong | II |
| IP109 | Amneal Pharma | 5 mg | 325 mg | White oblong | II |
| T257 | Camber Pharma | 5 mg | 325 mg | White oblong | II |
The M366 is the only Mallinckrodt hydrocodone tablet that is genuinely oval rather than oblong. A patient who receives an M367 believing it to be an M365 has received double the hydrocodone dose. Counterfeiters exploit this confusion by pressing high-dose counterfeit tablets with lower-dose imprints to obscure the quantity of fentanyl incorporated.
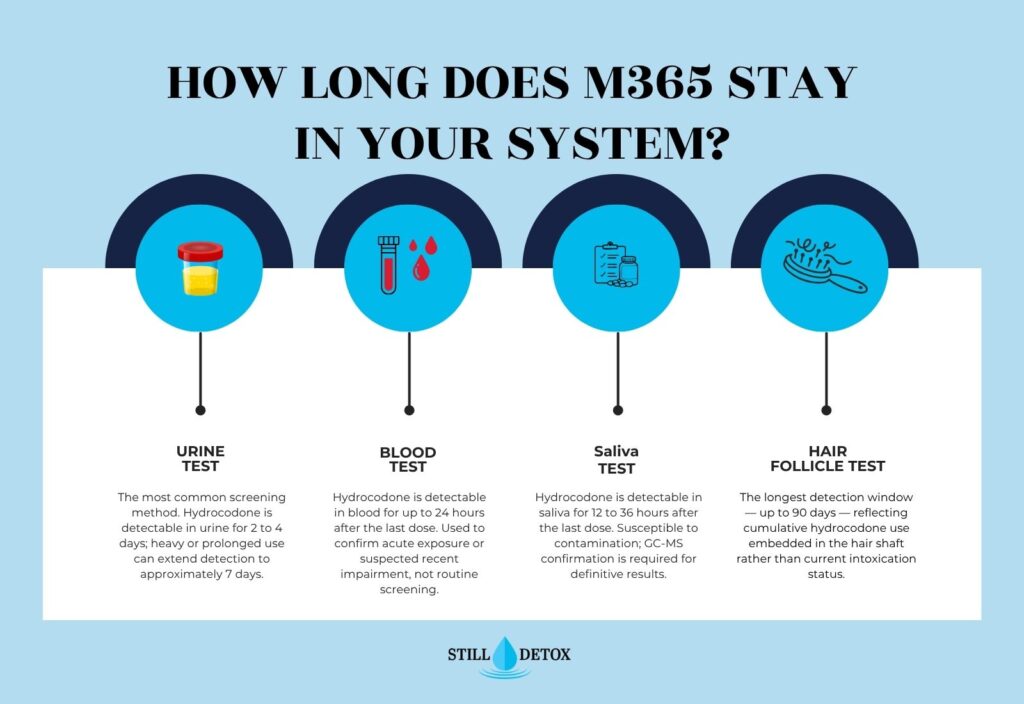
How Long Does the M365 Pill Stay in Your System?
Hydrocodone reaches peak plasma concentration approximately 1.3 hours after ingestion and produces analgesic effects within 30 to 60 minutes. The immediate-release half-life of approximately 3.8 hours determines the standard prescribed dosing interval of every four to six hours. CYP2D6 ultra-rapid metabolizers clear hydrocodone faster; poor metabolizers retain elevated plasma concentrations beyond the expected dosing window.
Hydrocodone detection windows by specimen type:
- Urine: 2 to 4 days after last dose; the most common screening matrix; heavy or prolonged use can extend detection to approximately 7 days
- Blood: up to 24 hours after last dose; used for acute exposure confirmation rather than routine screening
- Saliva: 12 to 36 hours after last dose; susceptible to environmental contamination
- Hair follicle: up to 90 days; reflects use history rather than current intoxication status
Standard opioid immunoassay panels may produce false-negative results for hydrocodone specifically. Gas chromatography-mass spectrometry (GC-MS) confirmation testing is required for definitive identification.
Side Effects and Risks of the M365 Pill
The M365 pill produces side effects through mu-opioid receptor activation across both the central and peripheral nervous systems, with risk severity scaling directly with dose, duration of use, and co-ingested substances.
Common Side Effects
Hydrocodone bitartrate activates mu-opioid receptors in the gastrointestinal enteric nervous system as well as the CNS, producing a predictable cluster of peripheral effects alongside analgesia. Most individuals experience at least one of the following at therapeutic doses:
Common side effects of the M365 pill include:
- Nausea and vomiting: driven by hydrocodone’s activation of the chemoreceptor trigger zone in the medulla oblongata, most pronounced with the first dose
- Constipation: caused by reduced intestinal peristalsis and increased anal sphincter tone; does not resolve with tolerance as most other side effects do
- Sedation and cognitive slowing: dose-dependent CNS depression that diminishes as tolerance develops over days to weeks of regular use
- Dizziness and orthostatic hypotension: resulting from opioid-induced vasodilation and blunted baroreceptor reflex responses
- Pruritus (itching): driven by histamine release from mast cells rather than an IgE-mediated allergic hypersensitivity reaction
- Miosis (pinpoint pupils): resulting from oculomotor nerve stimulation at the Edinger-Westphal nucleus; persists even with tolerance development
- Urinary retention and dry mouth: reflecting anticholinergic effects on smooth muscle tone and salivary gland function
Severe Side Effects and Overdose Warning Signs
Hydrocodone overdose suppresses the pre-Botzinger complex in the brainstem, eliminating the automatic breathing drive and producing apnea within minutes. Seek emergency medical care immediately if any of the following appear after M365 use:
- Breathing that is very slow (fewer than 8 breaths per minute), shallow, or has stopped entirely
- Pinpoint pupils that do not dilate in low light or complete darkness
- Unresponsiveness or inability to be woken by voice or sternal pressure
- Blue or gray discoloration of the lips, fingernails, or skin (cyanosis)
- Gurgling or choking sounds indicating airway obstruction from muscle limpness
- Complete limpness or total absence of muscle tone
- Seizures, particularly if acetaminophen toxicity or fentanyl contamination is suspected
Naloxone (Narcan) competitively displaces hydrocodone and hydromorphone from mu-opioid receptors, reversing respiratory depression within 2 to 5 minutes. Administer 4 mg intranasal naloxone and call 911 simultaneously; administer a second dose after 2 to 3 minutes if breathing does not improve. Counterfeit M365 pills containing fentanyl or carfentanil require multiple naloxone doses because their higher mu-receptor binding affinity can outlast a single 4 mg administration.
Acetaminophen toxicity follows a separate, delayed pathway: N-acetyl-p-benzoquinone imine (NAPQI) depletes hepatic glutathione stores and binds hepatocyte proteins, producing acute liver failure 72 to 96 hours after ingestion. N-acetylcysteine (NAC) is the antidote and achieves maximum hepatoprotective effect when initiated within 8 to 10 hours of ingestion.
Polydrug use amplifies M365 overdose risk through mechanistic synergy: opioids suppress the brainstem respiratory drive while benzodiazepines suppress the compensatory arousal response. This dual-pathway asphyxiation mechanism prompted the FDA’s 2016 black box warning against concurrent opioid and benzodiazepine prescribing.
Long-Term Effects of M365 Misuse
Sustained hydrocodone misuse produces neuroadaptive changes that outlast the period of active use, altering pain sensitivity, hormonal function, and immune competence. CYP2D6 enzyme induction and progressive mu-opioid receptor downregulation collectively remodel the brain’s reward and stress-response architecture over months of continuous exposure.
Long-term consequences of M365 misuse include:
- Opioid-induced hyperalgesia (OIH): paradoxical increased pain sensitivity from sustained mu-receptor activation; patients experience progressively more pain as hydrocodone doses increase, rather than less
- Post-acute withdrawal syndrome (PAWS): intermittent dysphoria, cravings, insomnia, and cognitive impairment persisting 4 to 24 months after cessation; represents the highest-risk window for relapse without treatment support
- Hypothalamic-pituitary-adrenal axis suppression: reduced testosterone and cortisol production causing hormonal dysregulation and sexual dysfunction
- Immune system dysregulation: opioid-induced inhibition of natural killer cell and T-cell function increasing vulnerability to bacterial and viral infection
- Neonatal abstinence syndrome (NAS): opioid-dependent newborns born to mothers who used M365 during pregnancy require medically supervised withdrawal management in a neonatal intensive care unit
Chronic high-dose M365 use elevates hepatic enzymes (AST, ALT) through acetaminophen accumulation, progressing to hepatic fibrosis in individuals with concurrent alcohol use disorder or pre-existing liver disease.
Counterfeit M365 Pills and Fentanyl Contamination
Counterfeit M365 tablets carry a life-threatening risk that is entirely independent of authentic hydrocodone pharmacology.
How Counterfeit M365 Tablets Are Manufactured
Counterfeit M365 pills are produced using commercial pill presses fitted with steel dies that replicate the authentic tablet’s 15 mm oblong profile, centered score line, and “M365” imprint. These operations produce up to 100,000 fake tablets per 12-hour shift, according to law enforcement seizure records. Counterfeit tablets substitute hydrocodone with fentanyl, para-fluorofentanyl, carfentanil, protonitazene, metonitazene, or xylazine, alone or in combination.
A lethal dose of fentanyl is approximately 2 milligrams. DEA laboratory analysis found individual counterfeit pills containing between 0.02 mg and 5.1 mg of fentanyl; the upper bound represents more than twice a lethal dose in a single tablet. Counterfeit pills laced with fentanyl drove approximately 73,000 synthetic opioid overdose deaths in the United States in 2023 alone.
Real vs. Fake M365: Visual Identification Guide
The DEA states that trained law enforcement personnel cannot reliably distinguish authentic M365 tablets from high-quality counterfeits without laboratory analysis. Visual inspection alone is not a safe verification method. However, seized counterfeit tablets frequently display manufacturing defects not present in pharmaceutical-grade production:
Visual warning signs in a potentially counterfeit M365 pill:
- Color irregularities: off-white, yellowish, or speckled coloring compared to the uniform clinical white of authentic Mallinckrodt tablets
- Imprint defects: shallow, smudged, off-center, or unevenly spaced “M365” characters that lack the clean, consistent depth of pharmaceutical stamping
- Surface texture anomalies: chalky, powdery, or crumbling consistency compared to the firm, smooth surface of authentic compressed tablets
- Dimensional inconsistency: variable pill sizes or weights within a single batch, indicating imprecise pressing equipment
- Score line irregularities: off-center placement, uneven depth, or absent center line on the tablet face
The “chocolate chip cookie effect” applies to fentanyl distribution within counterfeit batches: fentanyl concentrates unevenly across pressed pills, so one tablet may contain a lethal dose while an adjacent pill from the same batch contains none. Testing one pill does not guarantee the safety of others from the same source.
Fentanyl Test Strips and Naloxone Administration
Fentanyl test strips (FTS) detect fentanyl contamination in dissolved pill samples with 96 to 100% sensitivity. Testing protocol: crush the entire tablet, dissolve it in one-half teaspoon of water, dip the strip for 15 seconds, and read the result at 2 to 5 minutes. Two lines indicate a negative result; a single line on the left indicates fentanyl is present. A negative FTS result does not exclude carfentanil, nitazenes, or medetomidine, which standard immunoassay panels do not detect.
Xylazine test strips identify the veterinary sedative xylazine (street name: “tranq”), which does not respond to naloxone and requires emergency medical intervention independent of naloxone administration. If any opioid exposure is suspected in an unresponsive individual, administer naloxone immediately and call 911 without waiting for test strip results.
Hydrocodone Use Disorder and Withdrawal
Hydrocodone use disorder requires medically structured management because abrupt discontinuation triggers a withdrawal syndrome driven by noradrenergic rebound in the locus coeruleus.
Recognizing Hydrocodone Use Disorder
Hydrocodone use disorder (HUD) is a chronic neurobiological condition defined by the DSM-5 as compulsive opioid use persisting despite significant social, occupational, or health consequences. The DSM-5 specifies 11 diagnostic criteria across four domains: impaired control, social impairment, risky use, and pharmacological criteria (tolerance and withdrawal). Two to three criteria within 12 months indicate mild HUD; four to five indicate moderate HUD; six or more indicate severe HUD requiring intensive treatment.
Physical dependence on M365 develops through mu-opioid receptor downregulation and compensatory noradrenergic upregulation in the locus coeruleus, independent of whether DSM-5 criteria for HUD are met. Patients maintained on prescribed hydrocodone may develop physical dependence without meeting criteria for a use disorder. This distinction determines whether treatment planning requires supervised taper, MAT induction, or both.
Hydrocodone Withdrawal Timeline
Clinicians assess withdrawal severity using the Clinical Opiate Withdrawal Scale (COWS), an 11-item instrument scoring pulse rate, diaphoresis, restlessness, pupil size, bone and joint aches, rhinorrhea, GI upset, tremor, yawning, anxiety, and piloerection on a 0 to 47 scale. Scores of 5 to 12 indicate mild withdrawal; 13 to 24 indicate moderate withdrawal; scores above 36 indicate severe withdrawal requiring intensive clinical management and continuous monitoring.
Hydrocodone withdrawal progresses through five sequential phases:
- Hours 6 to 12: Anxiety, restlessness, yawning, lacrimation, and rhinorrhea emerge as locus coeruleus noradrenergic neurons reactivate without opioid suppression
- Hours 12 to 24: Myalgia, abdominal cramping, diaphoresis, piloerection, and insomnia intensify as sympathetic nervous system hyperactivity reaches its early peak
- Days 1 to 3: Nausea, vomiting, diarrhea, tachycardia, and hypertension reach maximum severity; dehydration from gastrointestinal fluid losses requires active clinical monitoring and rehydration
- Days 3 to 5: Acute somatic symptoms begin to resolve; persistent anhedonia, dysphoria, and insomnia mark the onset of post-acute withdrawal
- Weeks 2 to 24: PAWS produces intermittent cravings, mood instability, cognitive slowing, and sleep disruption; relapse risk during this window is highest without structured treatment support
Medication-Assisted Treatment for Hydrocodone Use Disorder
Medication-assisted treatment (MAT) combines FDA-approved pharmacotherapy with behavioral therapy to reduce withdrawal severity, suppress cravings, and prevent fatal relapse during the high-vulnerability post-acute period. Three pharmacotherapies carry FDA approval for opioid use disorder:
FDA-approved medications for hydrocodone use disorder:
- Buprenorphine/naloxone (Suboxone): a partial mu-opioid agonist with a ceiling effect limiting respiratory depression; initiated when COWS score reaches 8 to 12 to prevent precipitated withdrawal; reduces illicit opioid use and overdose mortality by more than 50% compared with no pharmacotherapy
- Naltrexone (Vivitrol injectable): a mu-opioid receptor antagonist that blocks hydrocodone-induced euphoria if relapse occurs; administered monthly as an extended-release intramuscular injection; requires 7 to 10 days of complete opioid abstinence before induction
- Methadone: a full mu-opioid agonist dispensed exclusively through federally licensed opioid treatment programs (OTPs); indicated for severe or treatment-refractory HUD where partial agonist therapy has not produced adequate outcomes
Behavioral therapies integrated with all MAT protocols include cognitive behavioral therapy (CBT), contingency management, and motivational interviewing, addressing the psychological reinforcement dimensions of HUD that pharmacotherapy alone does not resolve.
[IN-BLOG IMAGE 2: Insert Slide 3 image above the Treatment section below]
Hydrocodone Dependence Treatment at Still Detox
Still Detox provides a structured clinical continuum for individuals who develop physical dependence or hydrocodone use disorder through M365 use.
1- Medical Detox for Hydrocodone Withdrawal
Medical detox at Still Detox provides 24-hour clinical supervision throughout the acute hydrocodone withdrawal phase, monitoring COWS score progression, managing fluid replacement, and administering comfort medications to reduce withdrawal severity. The clinical team initiates buprenorphine/naloxone at appropriate COWS thresholds to minimize withdrawal intensity and address cravings before early discharge risk rises.
2- Residential Treatment for Opioid Use Disorder
Residential treatment at Still Detox addresses the neurobiological and behavioral dimensions of hydrocodone use disorder through daily structured programming that includes individual therapy, CBT groups, relapse prevention, and MAT continuation as clinically indicated. Same-day clinical assessments are available for individuals seeking immediate evaluation and admission.
Frequently Asked Questions
What is the M365 pill?
The M365 pill is a white oblong tablet containing hydrocodone bitartrate 5 mg and acetaminophen 325 mg, manufactured by Mallinckrodt Pharmaceuticals under DEA Schedule II classification. It is the generic equivalent of Norco 5/325. Doctors prescribe it for moderate to moderately severe acute pain that has not responded adequately to non-opioid analgesics.
How many mg of hydrocodone is in an M365 pill?
Each M365 tablet contains 5 mg of hydrocodone bitartrate paired with 325 mg of acetaminophen. Standard dosing is one to two tablets every four to six hours as prescribed. Exceeding 12 tablets in 24 hours crosses the FDA acetaminophen safety ceiling of 4,000 mg daily and increases hepatotoxicity risk significantly.
Is M365 the same as Norco?
Yes. The M365 pill is the generic equivalent of Norco 5/325, containing identical active ingredients at the same milligram doses. Norco is no longer available as a branded product. The M365 is not equivalent to Vicodin, which contained 300 mg of acetaminophen per tablet rather than the 325 mg in M365.
Is the M365 pill a controlled substance?
Yes. The DEA reclassified all hydrocodone combination products from Schedule III to Schedule II effective October 6, 2014, citing confirmed high abuse potential and severe dependence risk. Schedule II status means M365 prescriptions cannot be refilled; each dispensing requires a new written or electronic prescription from a licensed provider.
What does the M365 pill do to your body?
Hydrocodone in the M365 tablet binds mu-opioid receptors in the brain and spinal cord, blocking ascending pain signals and releasing dopamine in the limbic system. Acetaminophen independently inhibits COX enzyme activity in the CNS, adding analgesia through a separate pathway. At doses exceeding the prescription or in CYP2D6 ultra-rapid metabolizer individuals, respiratory depression becomes a life-threatening risk.
How long does M365 stay in your system?
Hydrocodone has an immediate-release half-life of approximately 3.8 hours. Detection windows are 2 to 4 days in urine, up to 24 hours in blood, 12 to 36 hours in saliva, and up to 90 days in hair follicles. Heavy or prolonged use can extend urine detection to approximately 7 days beyond the last dose.
Can you overdose on M365?
Yes. Hydrocodone overdose suppresses brainstem respiratory centers, causing breathing to slow or stop entirely. Counterfeit M365 pills containing fentanyl dramatically elevate overdose risk. If someone becomes unresponsive, breathes abnormally, or presents with pinpoint pupils after M365 use, administer naloxone immediately and call 911.
What is the difference between M365 and M367?
Both the M365 and M367 are white oblong tablets manufactured by Mallinckrodt Pharmaceuticals, each containing 325 mg of acetaminophen. The key difference is hydrocodone dose: M365 contains 5 mg; M367 contains 10 mg, making it twice as potent. Both carry identical Schedule II classification and equivalent counterfeit risk.
References
- Drug Enforcement Administration. (2014, August 21). DEA to publish final rule rescheduling hydrocodone combination products. U.S. Department of Justice. https://www.dea.gov/press-releases/2014/08/21/dea-publish-final-rule-rescheduling-hydrocodone-combination-products
- U.S. National Library of Medicine. (2025). Hydrocodone bitartrate and acetaminophen tablet [DailyMed drug label, Mallinckrodt Inc.]. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=c8be6c8d-7c55-459b-bc16-7651c2fd6e27
- Substance Abuse and Mental Health Services Administration. (2024). Results from the 2023 National Survey on Drug Use and Health: Detailed tables. U.S. Department of Health and Human Services. https://www.samhsa.gov/data/report/2023-nsduh-annual-national-report
- Drug Enforcement Administration. (2025). One Pill Can Kill. U.S. Department of Justice. https://www.dea.gov/onepill
- Food and Drug Administration. (2016). FDA drug safety communication: FDA warns about serious risks and death when combining opioid pain or cough medicines with benzodiazepines. U.S. Department of Health and Human Services. https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-warns-about-serious-risks-and-death-when-combining-opioid-pain-or
- National Institute on Drug Abuse. (2024). Opioid overdose crisis. U.S. Department of Health and Human Services. https://nida.nih.gov/research-topics/opioids/opioid-overdose-crisis
- Trescot, A. M., Datta, S., Lee, M., & Hansen, H. (2008). Opioid pharmacology. Pain Physician, 11(2 Suppl), S133–S153.
- Wesson, D. R., & Ling, W. (2003). The Clinical Opiate Withdrawal Scale (COWS). Journal of Psychoactive Drugs, 35(2), 253–259.
- American Psychiatric Association. (2022). Diagnostic and statistical manual of mental disorders (5th ed., text rev.). American Psychiatric Publishing.
- Gudin, J., & Fudin, J. (2020). A narrative pharmacological review of buprenorphine: A unique opioid for the treatment of chronic pain and opioid use disorder. Pain and Therapy, 9(1), 41–54.