Lyrica (pregabalin) is not an opioid. It belongs to a drug class called gabapentinoids, which work differently from opioids and do not bind to opioid receptors in the brain. Despite this, Lyrica is a Schedule V controlled substance because it can produce euphoric effects and carries a recognized risk of misuse.
Understanding exactly what drug class Lyrica falls into, why the DEA regulates it, and how it interacts with true opioids can help patients, caregivers, and those in recovery make safer, more informed decisions.
Key Takeaways
- Lyrica (pregabalin) is NOT an opioid or narcotic. It is an anticonvulsant (gabapentinoid) that works by calming overactive nerve signals, not by activating opioid receptors.
- Lyrica is a Schedule V controlled substance under the DEA’s Controlled Substances Act, the lowest federal schedule, due to its potential to cause euphoria and dependence.
- According to FDA clinical studies, approximately 23% of patients experience drowsiness and 30% experience dizziness as side effects of pregabalin.
- Combining Lyrica with opioids, benzodiazepines, or alcohol significantly increases the risk of respiratory depression and can be life-threatening.
- Lyrica is federally controlled in all U.S. states. Gabapentin (Neurontin), its close relative, is not federally scheduled, though several states regulate it independently.
What Is Lyrica (Pregabalin)?
Lyrica is the brand name for pregabalin, a prescription medication originally FDA-approved in 2004 as an anticonvulsant. It is available as immediate-release capsules, an oral solution, and an extended-release tablet called Lyrica CR.
Pregabalin belongs to the gabapentinoid drug class, which also includes gabapentin (Neurontin). Both drugs modulate calcium channel activity in the nervous system. They share structural similarities but are not interchangeable, and their legal classifications differ significantly.
Is Lyrica an Opioid or Narcotic?
No. Lyrica is not an opioid and is not a narcotic in any clinical, pharmacological, or legal sense. Opioids work by binding to mu-opioid receptors in the brain to produce analgesia and euphoria. Pregabalin does not engage those receptors at all.
The confusion arises because Lyrica is a controlled substance that treats pain, which leads many people to assume it falls into the opioid or narcotic category. This assumption is incorrect. Lyrica targets nerve pain through a completely separate mechanism and is not related to drugs like oxycodone, hydrocodone, or morphine.
It also differs from other commonly misunderstood drugs. For example, is ketamine an opioid is another frequently searched question. Like Lyrica, ketamine is a controlled substance with pain-relieving properties that does not operate via opioid pathways.
What Drug Class Is Lyrica?
Lyrica is classified as a gamma-aminobutyric acid (GABA) analog and is grouped under anticonvulsants, also called antiepileptics. Within that broader class, pregabalin and gabapentin are specifically referred to as gabapentinoids.
Lyrica Drug Classification at a Glance
| Category | Classification |
|---|---|
| Drug Class | Gabapentinoid / Anticonvulsant |
| Generic Name | Pregabalin |
| Brand Name | Lyrica, Lyrica CR |
| DEA Schedule | Schedule V (C-V) |
| Opioid? | No |
| Narcotic? | No |
| Federally Controlled? | Yes |
How Does Lyrica Work?
Pregabalin binds to the alpha-2-delta subunit of voltage-gated calcium channels in the central nervous system. This binding reduces the release of excitatory neurotransmitters, including glutamate, norepinephrine, and substance P.
By dampening these signals, Lyrica decreases abnormal pain transmission from damaged nerves and lowers the frequency of epileptic discharges. It does not act on opioid receptors, dopamine receptors, or serotonin receptors. Its mechanism is distinct from opioids, benzodiazepines, and antidepressants.
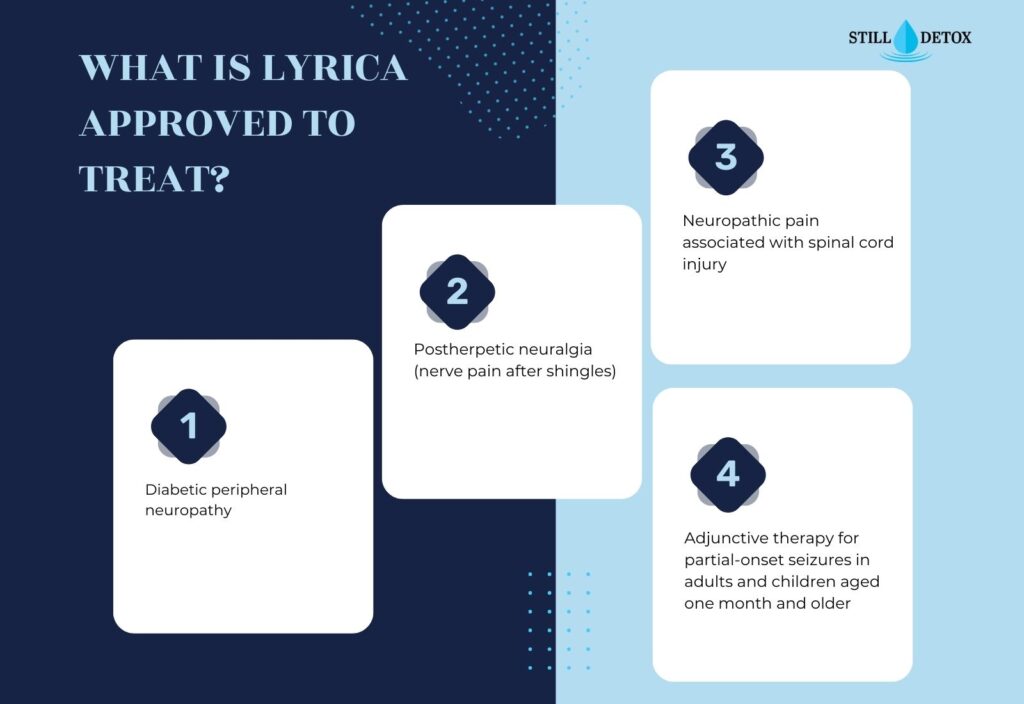
What Is Lyrica Approved to Treat?
The FDA has approved pregabalin for five specific indications. Clinicians may also use it off-label for additional conditions, though the evidence base varies.
FDA-approved uses include:
- Diabetic peripheral neuropathy
- Postherpetic neuralgia (nerve pain after shingles)
- Fibromyalgia
- Neuropathic pain associated with spinal cord injury
- Adjunctive therapy for partial-onset seizures in adults and children aged one month and older
Lyrica is not a first-line opioid alternative for general acute pain. It targets neuropathic and seizure-related conditions specifically, making it distinct in purpose from traditional pain medications.
Why Is Lyrica a Controlled Substance?
The DEA placed Lyrica in Schedule V under the Controlled Substances Act after reviewing evidence that high doses can produce euphoria, altered perception, and intoxication. Some patients describe the effect as similar to alcohol or marijuana, including feelings of calm, excitement, or happiness.
Schedule V represents the lowest federal control level. It indicates that the drug has accepted medical use and limited but real abuse potential. Compared to Schedule II opioids like oxycodone, Lyrica has milder withdrawal symptoms and a lower overdose risk on its own. However, misuse does occur, particularly among individuals with a substance use history.
Because it is a federally controlled substance, pharmacies must track Lyrica dispensing. Prescribers are required to consult state prescription drug monitoring programs (PDMPs) before issuing refills, and quantities are limited to prevent excessive prescriptions.
Why Is Lyrica a Controlled Substance and Neurontin Is Not?
This is one of the most frequently searched questions about these two drugs, and the answer comes down to abuse potential data. When the FDA reviewed pregabalin before approving it, clinical trial data showed a higher frequency of euphoria-related adverse events compared to gabapentin.
Gabapentin is not scheduled at the federal level, though states including Kentucky, Tennessee, Virginia, Michigan, and West Virginia have independently classified it as a controlled substance. Several other states require gabapentin to be tracked under prescription monitoring programs.
The structural similarity between the two drugs leads many to assume they carry the same legal status. They do not. If you are managing withdrawal or tapering from either medication, reviewing gabapentin withdrawal symptoms and timeline provides useful clinical context.
Lyrica vs. Gabapentin: Key Differences
| Feature | Lyrica (Pregabalin) | Gabapentin (Neurontin) |
|---|---|---|
| Drug Class | Gabapentinoid | Gabapentinoid |
| Federally Controlled? | Yes, Schedule V | No (some states control it) |
| Time to Peak Concentration | ~1 hour | 3 to 4 hours |
| Bioavailability | High, predictable (~90%) | Variable (30% to 60%) |
| Fibromyalgia Approval | Yes | No |
| Spinal Cord Injury Pain Approval | Yes | No |
| Abuse Potential Data | Higher (federal scheduling) | Lower (no federal scheduling) |
Both drugs belong to the same class and share a mechanism of action, but pregabalin is more potent, more predictable in absorption, and more tightly regulated as a result.
Is Lyrica Addictive?
Lyrica carries a real but lower addiction risk compared to opioids or benzodiazepines. Physical dependence can develop with regular use, especially at higher doses, and abrupt discontinuation may cause withdrawal symptoms, including insomnia, headaches, nausea, and anxiety.
People with a prior history of substance use disorder face a significantly elevated risk of developing dependence on pregabalin. Some individuals misuse Lyrica specifically to enhance the effects of opioids or benzodiazepines, which compounds both the addiction risk and the danger of overdose.
Understanding Lyrica addiction and mixing risks in detail is important for patients on long-term pregabalin therapy, especially those with any history of substance use.
Dangers of Mixing Lyrica with Opioids or Benzodiazepines
This is where the real safety risk lies. Lyrica is a mild CNS depressant on its own, but when combined with other depressants, particularly opioids or benzodiazepines, the interaction can be fatal.
Combining pregabalin with drugs like oxycodone, hydrocodone, heroin, or fentanyl significantly magnifies respiratory depression. The FDA requires a black box warning on Lyrica stating that concomitant use with opioids can cause serious respiratory depression, coma, and death. Similarly, combining Lyrica with benzodiazepines raises the risk of excessive sedation. For patients managing opioid treatment or recovery, a properly managed benzodiazepine taper schedule can minimize the overlap of CNS depressants.
The threat is not limited to prescription medications. The rise of counterfeit pills containing fentanyl and other synthetic opioids makes accidental mixing increasingly dangerous for anyone taking any CNS-active medication, including Lyrica.
Frequently Asked Questions
What class of drug is pregabalin?
Pregabalin belongs to the gabapentinoid class of anticonvulsants, also known as antiepileptics. It is a GABA analog that works by binding to calcium channels in the central nervous system. It is not an opioid, benzodiazepine, NSAID, or antidepressant. The DEA classifies it as a Schedule V controlled substance due to its documented euphoric potential at high doses.
What is considered an opioid?
An opioid is any drug that binds to and activates opioid receptors in the brain, spinal cord, and other tissues to produce analgesia. This category includes natural opiates like morphine and codeine, semi-synthetic opioids like oxycodone and hydrocodone, and fully synthetic opioids like fentanyl and methadone. Lyrica does not activate opioid receptors and is not considered an opioid.
Is pregabalin a very strong painkiller?
Yes, Pregabalin provides significant relief for neuropathic pain conditions, with many patients noticing improvement within the first week of treatment. It is not effective for acute or musculoskeletal pain in the way that opioids or NSAIDs are. Its strength is specific to nerve-related pain. Doses typically range from 75 mg to 300 mg per day, titrated based on tolerability.
Is gabapentin an opioid medication?
No. Gabapentin (Neurontin) is also a gabapentinoid anticonvulsant and is not an opioid. Like pregabalin, it does not act on opioid receptors. It is not federally scheduled in the U.S., though several states have classified it as a controlled substance due to documented misuse patterns and its ability to enhance opioid euphoria when combined.
Can you overdose on Lyrica?
Yes, a Lyrica overdose is possible, especially when combined with opioids, alcohol, or benzodiazepines. Symptoms include extreme drowsiness, confusion, slowed breathing, and loss of consciousness. The FDA requires the Lyrica label to carry a warning specifically about the risk of fatal respiratory depression when taken alongside opioid medications.
Is Lyrica a painkiller?
Yes, Lyrica is used to manage certain types of pain, specifically neuropathic pain caused by conditions like diabetic neuropathy, shingles, fibromyalgia, and spinal cord injury. It is not a conventional painkiller like ibuprofen or acetaminophen and does not treat acute injury pain. Its mechanism targets abnormal nerve signaling rather than inflammation or opioid pathways.
References
- Drug Enforcement Administration. (2024). Drug scheduling. U.S. Department of Justice. https://www.dea.gov/drug-information/drug-scheduling
- Food and Drug Administration. (2024). Lyrica (pregabalin) prescribing information. U.S. Department of Health and Human Services. https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/021446s038lbl.pdf
- National Library of Medicine. (2024). Pregabalin. MedlinePlus. https://medlineplus.gov/druginfo/meds/a605045.html
- Pope, C. (2025, July 25). Pregabalin (Lyrica). Drugs.com. (APA citation only, no external link)