Tramadol withdrawal symptoms can begin within 8 to 24 hours after the last dose and range from flu-like discomfort to severe anxiety, insomnia, and neurological effects. Because tramadol acts on both opioid receptors and monoamine neurotransmitters, its withdrawal pattern is more complex than most prescription painkillers. Understanding what to expect and when to seek medical support can mean the difference between a managed detox and a dangerous one.
Key Takeaways
- Tramadol withdrawal symptoms typically begin 8 to 24 hours after the last dose and resolve within 5 to 10 days for most people.
- Approximately 1.7 million Americans are estimated to be physically dependent on tramadol, according to SAMHSA data.
- Around 90% of people in tramadol withdrawal experience typical opioid-type symptoms; roughly 10% experience atypical neurological effects, including hallucinations and panic attacks.
- Stopping tramadol cold turkey carries a risk of seizures, particularly at higher doses or after long-term use.
- Medically supervised detox with a structured tapering plan is the safest and most effective route to managing tramadol withdrawal.
What Is Tramadol and Why Does Withdrawal Happen?
Tramadol is a Schedule IV synthetic opioid prescribed for moderate to moderately severe pain. Unlike traditional opioids, it works through two mechanisms. It binds to opioid receptors in the brain and simultaneously inhibits the reuptake of serotonin and norepinephrine throughout the nervous system.
This dual action is what makes tramadol withdrawal unique. The brain adapts to the constant presence of tramadol by adjusting its own chemistry. When the drug is removed, both the opioid and serotonin-norepinephrine systems go into a state of imbalance, producing withdrawal symptoms that can feel more layered and intense than other opioid discontinuations.
Physical dependence can develop even when tramadol is taken exactly as prescribed. The U.S. Food and Drug Administration has placed black box warnings on tramadol formulations, noting the risks of addiction, misuse, and serious withdrawal reactions upon abrupt discontinuation.
If you are comparing tramadol to other pain medications, understanding the differences between Toradol and tramadol can help clarify why tramadol carries dependence risk while non-opioid alternatives do not.
Tramadol Withdrawal Symptoms
Tramadol produces two distinct withdrawal syndromes depending on which receptor system is most affected. Most people experience the typical opioid-type syndrome. A smaller subset, around 10%, develop atypical symptoms tied to serotonin and norepinephrine disruption.
Typical Opioid Withdrawal Symptoms (Approximately 90% of Cases)
These are the most common tramadol withdrawal symptoms and closely resemble withdrawal from other short-acting opioids:
- Sweating and chills
- Muscle aches and body pain
- Restlessness and irritability
- Nausea, vomiting, and diarrhea
- Abdominal cramping
- Runny nose and watery eyes
- Insomnia and disrupted sleep
- Intense drug cravings
- Fatigue and low energy
- Rapid heartbeat
These symptoms are rarely life-threatening but can be physically exhausting and psychologically overwhelming, particularly for people managing chronic pain at the same time.
Atypical Withdrawal Symptoms (Approximately 10% of Cases)
Because tramadol also inhibits serotonin and norepinephrine reuptake, a minority of users experience symptoms that go beyond the standard opioid withdrawal pattern:
- Severe anxiety and panic attacks
- Hallucinations (visual or auditory)
- Paranoia and confusion
- Depersonalization or dissociation
- Numbness or tingling in the extremities
- Mood swings and emotional instability
- Psychosis (rare but documented)
These atypical symptoms are more likely in people who have been taking higher doses for longer periods, those with underlying mental health conditions, or individuals who have been combining tramadol with antidepressants or other serotonergic medications.
Using the Clinical Opiate Withdrawal Scale (COWS) can help clinicians objectively measure the severity of tramadol withdrawal and guide appropriate treatment decisions.
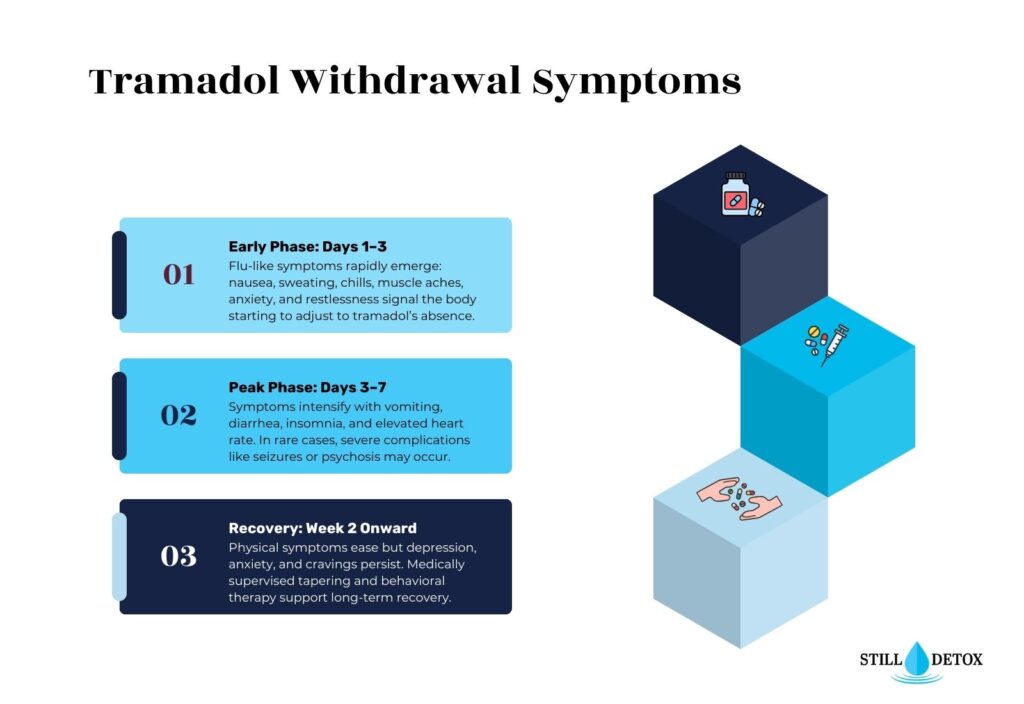
Tramadol Withdrawal Timeline
The tramadol withdrawal timeline is shaped by whether someone takes immediate-release or extended-release formulations, the dose, duration of use, and individual metabolism. The following is the typical progression for immediate-release tramadol.
- 8 to 24 Hours After Last Dose: Withdrawal begins. Early symptoms include anxiety, restlessness, sweating, and muscle discomfort. Cravings often appear during this window. Extended-release tramadol users may not notice symptoms for up to 36 hours due to the slower release mechanism.
- Days 1 to 3: Symptoms peak in intensity during this phase. Nausea, vomiting, diarrhea, insomnia, body aches, and chills are at their worst. For users experiencing atypical symptoms, panic attacks and hallucinations are most likely to emerge during this period. Medical supervision is most critical at this stage.
- Days 4 to 7: Physical symptoms begin to subside gradually. Stomach cramps, diarrhea, and body pain may persist but with reduced intensity. Psychological symptoms, including anxiety, depression, and irritability, often become more prominent as the acute physical phase winds down.
- Days 8 to 14: Most acute opioid-type symptoms resolve for the majority of people. However, mood disturbances, disrupted sleep, and low energy may continue. Those with a history of anxiety or depression may find this stage particularly difficult without psychiatric support.
- Beyond Two Weeks: Post-Acute Withdrawal Syndrome (PAWS) Some individuals experience persistent symptoms for weeks, months, or, in rare cases, longer. This is known as post-acute withdrawal syndrome. PAWS from tramadol can include chronic fatigue, depression, cognitive fog, and ongoing sleep problems. Research suggests that approximately 90% of individuals recovering from opioid dependence experience some degree of PAWS.
Factors That Affect Tramadol Withdrawal Severity
Not everyone experiences tramadol withdrawal the same way. Several variables influence how severe and how long symptoms last:
- Dose and Duration of Use: Higher doses taken over longer periods produce more pronounced withdrawal. Someone who took 50 mg daily for a few weeks will have a significantly milder experience than someone on 400 mg daily for years.
- Formulation Type: Immediate-release tramadol has a half-life of roughly 6 hours, so withdrawal starts quickly. Extended-release versions have a longer half-life, which delays onset but can prolong the overall process.
- Concurrent Medications: Combining tramadol with other CNS depressants or serotonergic drugs complicates withdrawal. The risks of mixing tramadol and gabapentin are well documented and add complexity to the discontinuation process.
- Individual Health Factors: Age, liver function, metabolic rate, and overall physical health all influence how quickly the body clears tramadol and how forcefully it rebounds.
- Mental Health History: Pre-existing anxiety, depression, or bipolar disorder significantly amplifies the psychological dimension of withdrawal. People in this category are more likely to experience atypical symptoms and PAWS.
- Method of Use: People who misused tramadol by snorting or taking it in ways other than prescribed tend to develop stronger dependence faster, which intensifies withdrawal. Understanding tramadol misuse patterns is important context for clinicians developing a detox plan.
Cold Turkey vs. Tapering: Which Is Safer?
Stopping tramadol abruptly is not recommended by most medical guidelines. Abrupt discontinuation at higher doses carries a meaningful risk of seizures, a danger that is less common with many other opioids due to tramadol’s effect on serotonin thresholds.
Tapering, which means gradually reducing the dose over a structured schedule, allows the brain and nervous system to adjust incrementally. A proper taper minimizes withdrawal intensity, reduces seizure risk, and makes the overall process more manageable. There is no universal tapering schedule. A physician will factor in the patient’s dose, duration of use, health status, and pain management needs when designing an individualized plan.
The general approach used by many clinicians involves a 10 to 25% dose reduction every one to four weeks, depending on tolerance. The taper may be paused or reversed temporarily if withdrawal becomes unmanageable. This is particularly relevant when someone also takes other medications with withdrawal risk. For context, similar tapering principles apply when discontinuing other CNS-active drugs, as seen in gabapentin withdrawal management.
Tramadol Detox Treatment and Medications
Medical detox is the first and most important step in recovering from tramadol dependence. A supervised setting provides around-the-clock monitoring, symptom management, and access to medications that can significantly reduce discomfort.
Medications Commonly Used in Tramadol Detox
- Buprenorphine (Suboxone): FDA-approved for opioid use disorder, buprenorphine reduces cravings and eases the physical symptoms of opioid withdrawal. It can be used during the detox phase and continued as maintenance medication.
- Methadone: Used in opioid treatment programs, methadone stabilizes patients during detox and can be tapered slowly over time. It is particularly useful for people with severe or prolonged dependence.
- Lofexidine (Lucemyra): An FDA-approved non-opioid medication that targets physical withdrawal symptoms such as sweating, muscle aches, and rapid heartbeat by reducing norepinephrine activity.
- Clonidine: Often used off-label to manage autonomic symptoms of opioid withdrawal including elevated heart rate, sweating, and anxiety.
- Supportive Medications: Anti-nausea medications, antidiarrheals, sleep aids, and non-opioid analgesics may be used symptomatically during the acute withdrawal phase.
The opioid withdrawal timeline more broadly outlines what medically managed opioid detox looks like, which is directly applicable to tramadol treatment planning.
After medical detox, ongoing treatment is essential. Detox alone is not sufficient for long-term recovery. Continued care may include inpatient residential treatment, outpatient therapy, medication-assisted treatment (MAT), behavioral therapies such as cognitive behavioral therapy (CBT), and peer support groups.
When to Seek Immediate Medical Help
Certain symptoms during tramadol withdrawal require emergency care. Do not attempt to manage the following at home:
- Seizures or loss of consciousness
- Severe confusion, hallucinations, or psychosis
- Chest pain or difficulty breathing
- Signs of serotonin syndrome: fever, agitation, rapid heart rate, muscle twitching, or incoordination
- Inability to keep down fluids leading to dehydration
Anyone stopping tramadol after long-term use at high doses, or anyone combining it with antidepressants, should pursue medically supervised detox rather than attempting to quit at home. The risk profile for tramadol is distinct enough from other opioids that home withdrawal carries real dangers that clinical support can prevent.
Frequently Asked Questions
How long do tramadol withdrawal symptoms last?
For most people, acute tramadol withdrawal lasts 5 to 10 days after the last dose. Physical symptoms like nausea, sweating, and muscle pain typically peak around days 2 to 3 and begin resolving by day 7. Psychological symptoms, including anxiety, depression, and insomnia, can persist for several weeks. Those with a history of heavy or prolonged use may experience PAWS beyond the acute phase.
How long does it take for tramadol side effects to wear off?
Tramadol’s short-term side effects, such as drowsiness, nausea, and dizziness, generally clear within 6 to 12 hours for immediate-release formulations as the drug exits the system. Withdrawal-related side effects that emerge after stopping the drug are a separate process and follow the withdrawal timeline outlined above, typically resolving fully within two weeks for most people.
What medication is used to detox from tramadol?
The most commonly used medications for tramadol detox include buprenorphine (Suboxone), methadone, and lofexidine (Lucemyra). Clonidine is also used off-label to manage autonomic withdrawal symptoms. The right medication depends on the severity of dependence, co-occurring conditions, and whether long-term MAT is part of the recovery plan. A physician should evaluate each case individually before initiating detox medication.
How to remove tramadol from the body?
The body clears tramadol primarily through the liver and kidneys. There is no medically validated method to accelerate this process. Staying well-hydrated, eating nutritiously, and getting adequate rest support the body’s natural clearance. The most effective and safest approach to removing tramadol from the body while managing withdrawal is a physician-supervised taper or medical detox program, not rapid detox or unsupported home attempts.
References
- U.S. Drug Enforcement Administration. (2024). Tramadol. Retrieved from https://www.deadiversion.usdoj.gov/drug_chem_info/tramadol.pdf
- U.S. National Library of Medicine. (n.d.). Opiate and opioid withdrawal. MedlinePlus. Retrieved from
- Substance Abuse and Mental Health Services Administration. (n.d.). Emergency department visits for adverse reactions involving the pain medication tramadol. Retrieved from https://www.samhsa.gov/data/sites/default/files/report_1965/ShortReport-1965.html
- U.S. Food and Drug Administration. (2010). Tramadol hydrochloride extended-release capsules prescribing information.
- Centers for Disease Control and Prevention. (n.d.). Pocket guide: Tapering opioids for chronic pain.
- World Health Organization. (2014). Tramadol: Update review report. Expert Committee on Drug Dependence.
- Dart, R. C., Cicero, T. J., Surratt, H. L., Rosenblum, A., Bartelson, B. B., & Adams, E. J. (2012). Assessment of the abuse of tapentadol immediate release: The first 24 months. Journal of Opioid Management, 8(6), 395-402.